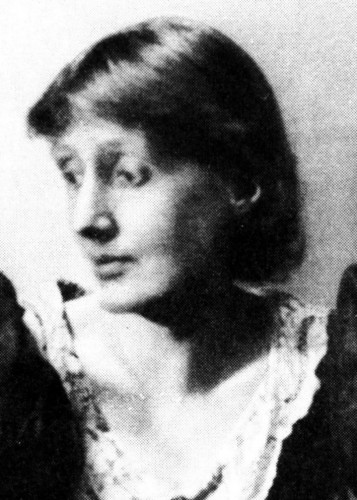Tehran, SAEDNEWS: Minoo Mohraz elaborated on the progress made on the 3rd phase of the clinical trial of the 'COV-Iran Barakat' vaccine at Eram Hotel on Sunday, saying: "With the mass production of the vaccine, we no longer need to import vaccines from abroad."
She said that the coronavirus outbreak had impacted the economic and educational aspects of the country, "so we must help everyone get vaccinated by October to reopen schools and universities."
In further remarks, Mohraz pointed out: "Everyone should be vaccinated," adding that given the shortage of vaccines globally, we must help people get vaccinated and get back to daily activities."
Mohraz noted: "COV-Iran Barakat's vaccine is effective and immune, and we hope that our vaccine would have the highest percentage of efficiency and immunity.”
Production capacity of 'COV-Iran Barakat' more than 10 million per month
Also, as the next keynote speaker, Hassan Jalili, the head of the vaccine production project in the Execution of Imam Khomeini's Order (EIKO), stated that the third phase of vaccine production would be launched in late spring.
Jalili said: "The production capacity of 'COV-Iran Barakat' is more than 10 million doses per month."
Speaking on Sunday at the press conference of COV-Iran Barkat vaccine, he said that the third phase includes industrial production, adding: "In fact, this phase will be mass production, and we predict that this site will be launched in late spring and the vaccine production capacity will be more than 10 million doses per month."
"We should not forget that the world has been ahead of us in vaccines developing because they have been affected by various viral diseases such as Severe acute respiratory syndrome (SARS) and Middle East Respiratory Syndrome (MERS)," Jalili noted.
He noted that Iran has now been able to indigenize and develop the vaccine.
"Our main concern since the first day of the clinical trial has been the mass production," he said, noting: "We can not wait for the results of phases one, two, and three to be completed and the results to be determined and then start mass production."
Of course, other countries have begun mass production of the vaccine as soon as the vaccine has been proven successful in the laboratory and clinical phase.
"We have designed the production of vaccines in the country in three phases," he said and stressed: "The first phase only provides the amount of vaccine for clinical study in phases one, two, and three."
"The semi-industrial phase or phase 2, which has been almost completed, includes the construction and installation of its equipment," Jalili added.
He underscored: "In the semi-industrial phase, our prediction is to produce between one and two million doses of vaccine per month."
Jalili stressed: "The project to make a vaccine is progressing according to the schedule, and there has been no delay."
Regarding other vaccine platforms that the EIKO is studying to enter the human phase, he added: "The DNA-based platforms have completed their preclinical phase, and the relevant documents will be submitted to the Ministry of Health within the next week to two weeks later to apply for a human study."
Jalili said: "So far, based on what has been seen in pre-clinical studies, the results have been satisfactory, but we have to wait for the relevant experts to comment on the documents provided, and we hope that this vaccine will be able to enter the human study phase."
He said other platforms are also conducting preclinical studies (Source: Iran Press).


 Minou Moharaz Distinguished Iranian Professor of Infectious Diseases Guarantees the Safety of COV-Iran
Minou Moharaz Distinguished Iranian Professor of Infectious Diseases Guarantees the Safety of COV-Iran